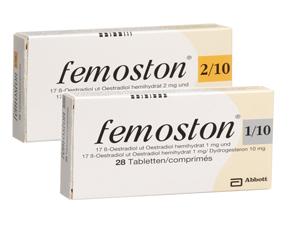
Femoston
Incl. online consultation, medicine and discreet delivery
View Options
The medical information on this site has been reviewed by Dr. Ross Elledge (GMC registered) and is provided for educational purposes. It does not replace a face-to-face consultation with your GP or specialist. Always follow the advice of your prescribing doctor and read the patient information leaflet supplied with your medication.
Choose Your Medicine
Select dosage and quantity
Online Consultation
Reviewed by a qualified physician
Fast, Discreet Delivery
Delivered to your door
Medical Information
About This Medicine
Femoston is a hormone replacement therapy (HRT) containing estradiol and dydrogesterone, available in both sequential and continuous combined formulations.
It is prescribed for the relief of menopausal symptoms and for the prevention of postmenopausal osteoporosis in women at risk of fracture when other treatments are not suitable.
Estradiol, the oestrogen component, replaces the natural hormone lost after the menopause, relieving symptoms such as hot flushes, night sweats, vaginal dryness, and mood changes.
Dydrogesterone is a progestogen that protects the endometrium from the proliferative effect of unopposed oestrogen, reducing the risk of endometrial hyperplasia and carcinoma.
Sequential vs Continuous Combined
Femoston 1/10 and 2/10 are sequential preparations: oestrogen is taken daily, with dydrogesterone added for the last 14 days of each 28-day cycle.
A withdrawal bleed typically occurs during the oestrogen-only phase. These are appropriate for perimenopausal women or those in early menopause.
Femoston Conti (1/5 or 0.5/2.5) provides both hormones daily without a break and is designed for women who are at least 12 months postmenopausal and wish to avoid monthly bleeds.
Usage & Dosage
How to Take Femoston
Take one tablet daily at the same time, with or without food. Swallow whole with water.
Sequential Formulations (1/10, 2/10)
Take the oestrogen-only tablets (white or red) for the first 14 days, then the combined tablets (grey or yellow) for the next 14 days. Start the next pack immediately without a break.
A withdrawal bleed may occur during the first half of each cycle.
Continuous Combined Formulation (Conti)
Take one combined tablet every day without interruption. No withdrawal bleed is expected, though some irregular spotting may occur in the first months.
Missed Dose
Take the missed tablet as soon as you remember. If more than 12 hours have passed, skip it and resume the next day. Missing tablets may cause breakthrough bleeding.
Sequential (Perimenopausal Women)
- Femoston 1/10: estradiol 1 mg daily + dydrogesterone 10 mg for days 15-28
- Femoston 2/10: estradiol 2 mg daily + dydrogesterone 10 mg for days 15-28
Continuous Combined (Postmenopausal Women)
- Femoston Conti 1/5: estradiol 1 mg + dydrogesterone 5 mg daily
- Femoston Conti 0.5/2.5: estradiol 0.5 mg + dydrogesterone 2.5 mg daily
General Principles
- Use the lowest effective dose for the shortest duration
- Review at least annually
- No specific dose adjustment for renal impairment; contraindicated in active liver disease
Side Effects
Common (up to 1 in 10 women)
- Headache or migraine
- Breast tenderness or pain
- Abdominal pain, nausea, or bloating
- Back pain
- Irregular bleeding or spotting
Uncommon (up to 1 in 100 women)
- Depression or mood swings
- Weight gain
- Vaginal discharge
- Dizziness
Rare (up to 1 in 1,000 women)
- Venous thromboembolism (DVT, PE)
- Gallbladder disease
- Liver function disturbance
Very Rare (fewer than 1 in 10,000 women)
- Arterial thromboembolism (stroke, MI)
- Breast cancer (risk increases with duration of use beyond 5 years)
Breakthrough bleeding that persists beyond the first 6 months on continuous combined Femoston should be investigated.
Warnings & Precautions
Thromboembolism
HRT is associated with a small increase in the risk of venous thromboembolism. Risk factors include obesity, immobility, personal or family history of VTE, and thrombophilia.
Discontinue and seek urgent medical help if you develop sudden leg swelling, chest pain, or breathlessness.
Breast Cancer
Combined HRT is associated with a modestly increased risk of breast cancer, rising with duration of use. Attend all routine mammography appointments and report new breast lumps or changes promptly.
Endometrial Monitoring
Unexpected vaginal bleeding during continuous combined therapy or at times other than the expected withdrawal bleed on sequential therapy should be investigated.
Annual Review
The benefits and risks of continued HRT should be reassessed at least once a year. Use the lowest effective dose for the shortest necessary duration.
Contraindications
Do not take Femoston if you have:
- Known or suspected breast cancer (current or past)
- Known or suspected oestrogen-dependent malignancy
- Undiagnosed abnormal vaginal bleeding
- Untreated endometrial hyperplasia
- Active or recent venous or arterial thromboembolism
- Active liver disease or where liver function tests remain abnormal
- Known thrombophilic disorders (e.g., protein C, protein S, or antithrombin deficiency)
- Porphyria
- Hypersensitivity to estradiol, dydrogesterone, or any excipient
Frequently Asked Questions
What is the difference between Femoston sequential and Femoston Conti?
Will I have a period on Femoston Conti?
How long can I take Femoston?
Does Femoston help vaginal dryness?
Can I take Femoston if I have had a hysterectomy?
Dr. Ross Elledge
Consultant Surgeon · Oral & Maxillofacial Surgery
Verified Healthcare Professional