Exelon
Incl. online consultation, medicine and discreet delivery
View Options
The medical information on this site has been reviewed by Dr. Ross Elledge (GMC registered) and is provided for educational purposes. It does not replace a face-to-face consultation with your GP or specialist. Always follow the advice of your prescribing doctor and read the patient information leaflet supplied with your medication.
Choose Your Medicine
Select dosage and quantity
Online Consultation
Reviewed by a qualified physician
Fast, Discreet Delivery
Delivered to your door
Medical Information
About This Medicine
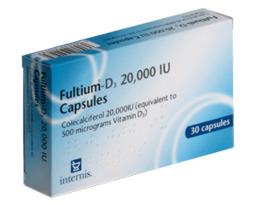
Exelon contains rivastigmine, a cholinesterase inhibitor prescribed for the symptomatic treatment of mild to moderately severe Alzheimer's disease and mild to moderately severe dementia associated with Parkinson's disease.
It is available as capsules, oral solution, and transdermal patches.
In Alzheimer's disease and Parkinson's disease dementia, there is a progressive loss of cholinergic neurones in the brain, leading to reduced levels of the neurotransmitter acetylcholine.
This deficit contributes to the cognitive impairment, memory loss, and behavioural changes characteristic of these conditions.
Rivastigmine inhibits both acetylcholinesterase and butyrylcholinesterase, the enzymes that break down acetylcholine, thereby increasing its availability at the synapse.
The transdermal patch formulation offers a continuous, smooth delivery of the drug over 24 hours, which results in fewer gastrointestinal side effects compared with oral dosing.
It also simplifies administration for carers, as a patch needs to be applied only once daily.
Clinical trials have demonstrated that rivastigmine provides modest but clinically meaningful improvements in cognition, global function, and activities of daily living.
Usage & Dosage
For oral capsules, take with food in the morning and evening. Swallow capsules whole with water.
For the transdermal patch, apply one patch daily to a clean, dry, hairless area of skin on the upper or lower back, upper arm, or chest. Press firmly for 30 seconds.
Replace the patch at approximately the same time each day and rotate the application site to avoid skin irritation. Remove the old patch before applying a new one.
The dose is increased gradually to the target dose over several weeks, as tolerated.
A carer should oversee administration and report any significant gastrointestinal symptoms, agitation, or changes in behaviour to the prescribing doctor.
Do not stop rivastigmine abruptly without medical advice.
Oral: start at 1.5 mg twice daily, increasing in steps of 1.5 mg twice daily at intervals of at least two weeks, up to a maximum of 6 mg twice daily. Patches: start with the 4.
6 mg/24 h patch; after at least four weeks, increase to the 9.5 mg/24 h patch if tolerated. A 13.3 mg/24 h patch may be used in patients with demonstrated benefit on 9.5 mg/24 h.
Dose reductions may be needed in hepatic impairment. Caution in renal impairment, though no formal adjustment is required.
Side Effects
Common side effects (more than 1 in 10 patients) include nausea, vomiting, diarrhoea, loss of appetite, and dizziness, particularly during dose escalation.
These effects are less frequent with the patch than with oral formulations.
Uncommon effects include headache, abdominal pain, weight loss, agitation, confusion, urinary incontinence, and tremor.
Skin reactions at the patch site (redness, itching, swelling) are common with the transdermal route.
Serious but rare adverse effects include severe vomiting leading to oesophageal rupture (particularly if the dose is restarted at the previous level after an interruption), bradycardia, atrioventricular block, seizures, gastrointestinal ulceration, and pancreatitis.
If treatment is interrupted for more than three days, it must be restarted at the lowest dose and re-titrated.
Warnings & Precautions
If treatment is interrupted for more than three consecutive days, rivastigmine must be restarted at the lowest dose and titrated upward again to avoid severe gastrointestinal reactions.
Monitor body weight regularly, as cholinesterase inhibitors can cause significant weight loss.
Use with caution in patients with sick sinus syndrome, conduction disturbances, active peptic ulcer disease, asthma, or COPD.
Only one patch should be worn at any time; accidental application of multiple patches has caused overdose.
Contraindications
Exelon is contraindicated in patients with known hypersensitivity to rivastigmine, other carbamate derivatives, or any excipient.
The patch formulation is contraindicated in patients with a history of application-site reactions suggestive of allergic contact dermatitis.
Caution is required in severe hepatic impairment, where drug exposure may be significantly increased.
Frequently Asked Questions
Is the patch better than the capsules?
What should I do if the patch falls off?
Can Exelon reverse Alzheimer's disease?
Why must the dose be re-titrated after a break in treatment?
Can Exelon be used for Parkinson's disease dementia?
Dr. Ross Elledge
Consultant Surgeon · Oral & Maxillofacial Surgery
Verified Healthcare Professional